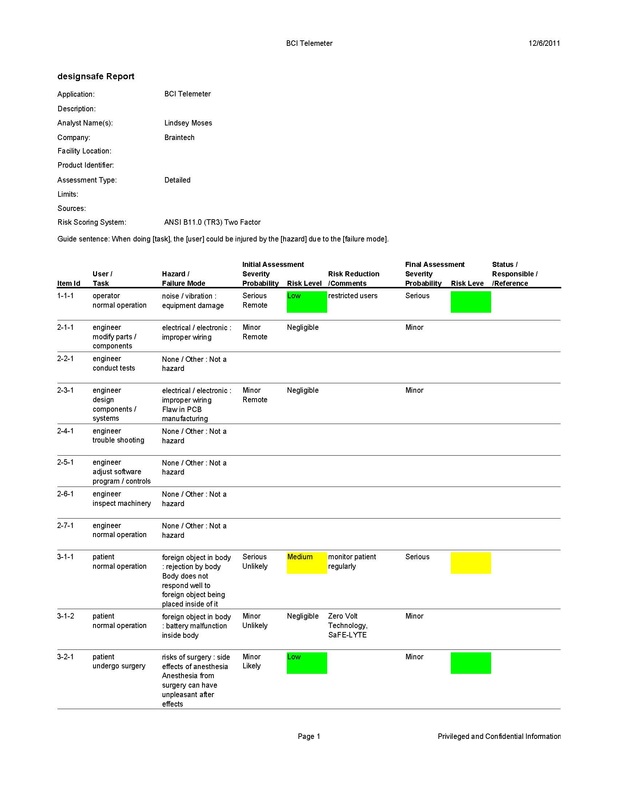
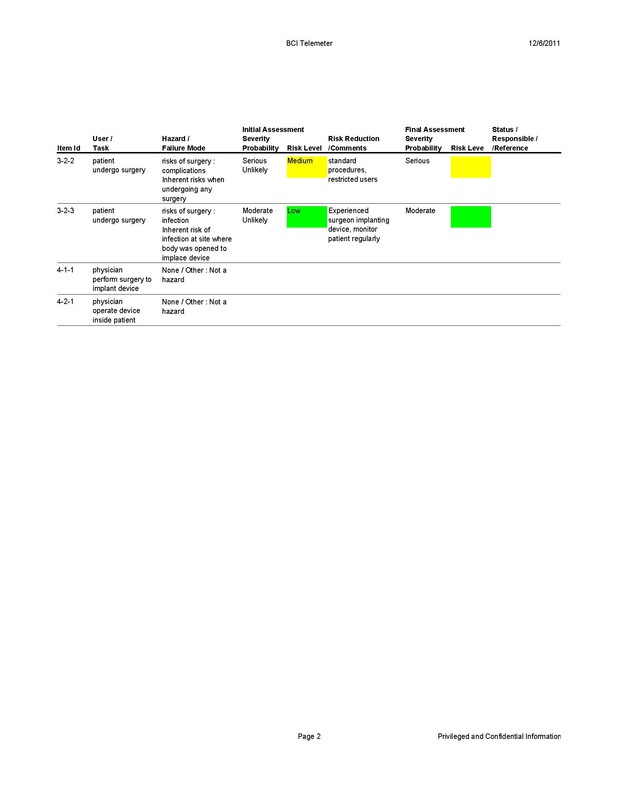
Safety Concerns
The BCI Telemeter poses a safety risk to the user since it must be surgically implanted into the body and remains there for an extended period of time. There is an initial risk to the patient due to the fact that invasive surgery is required to implant the ECoG electrode array on the dura of the brain and place the BCI Telemeter in the chest with internal wires connecting the two in order for the device to function. The specific risks of implanting the BCI Telemeter are the risks of undergoing any operation, as well as the risk of having a foreign object implanted in the body. The surgery for implantation is a standard procedure used to implant pacemakers and deep brain stimulators, and carries with it the risks of any surgery including side effects from anesthesia. Once the device has been implanted, the primary safety concern is the body’s reaction to the foreign object. To help alleviate this risk, the casing containing the device is coated in parylene, a biocompatible film that interacts safely with the internal environment of the body. The internal circuit components of the BCI Telemeter are contained in the hermetically sealed casing and will never come into direct contact with the body. The battery poses a few safety risks of its own that are accounted for by trademarked Zero Volt and SaFE-LYTE technologies provided by the Quallion battery. Zero Volt technology allows the battery to be stored for long periods of time with zero charge while still performing at peak capacity when in use, and SaFE-LYTE technology eliminates the risk of combustion of the electrolyte in the battery. The combination of these technologies makes it safe for the battery to be implanted and operate inside the body. A DesignSafe Analysis of the BCI Telemeter was performed and is shown below.